 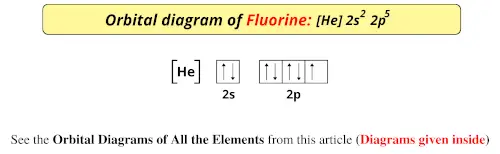
Also, one box can hold up to two electrons. 1s orbital contains 1 box, 2s orbital also contains 1 box, 2p orbital contains 3 boxes, 3s orbital contains 1 box, 3p orbital contains 3 boxes, and 4s orbital contains 1 box.Potassium has a total of 19 electrons and its electron configuration is 1s 22s 22p 63s 23p 64s 1.We know that potassium has a total of 19 electrons that need to be placed into orbitals, now for drawing its orbital diagram, we need to show its electrons in form of an arrow in different boxes using Aufbau, Hund’s, and Pauli’s exclusion rule. How to draw an Orbital diagram for Potassium? Let’s take one more example to understand the procedure of drawing the orbital diagram for an atom. These 3 electrons will go in the 2p orbital, since, the 2p orbital has 3 boxes, so, these electrons will be filled using Hund’s rule.Therefore, the first two electrons of nitrogen will go in the 1s orbital, and the next two will go into the 2s orbital, now we are left with 3 electrons.1s orbital contains 1 box, 2s orbital also contains 1 box and 2p orbital contains 3 boxes.As per the Aufbau rule, the electrons will be filled into 1s orbital first then 2s, then 2p…so on.Nitrogen has a total of 7 electrons and its electron configuration is 1s 22s 22p 3.We know that the nitrogen atom has a total of 7 electrons that need to be placed into orbitals, now for drawing its orbital diagram, we need to show its electrons in form of an arrow in different boxes using Aufbau, Hund’s, and Pauli’s exclusion rule.  
How to draw an Orbital diagram for Nitrogen? We have a diagonal rule for electron filling order in the different subshells using the Aufbau principle. This means that electrons can only enter higher-energy orbitals after lower-energy orbitals have been entirely filled.” “The Aufbau principle states that electrons will inhabit the lowest-energy orbitals first. The orbital diagram will be filled in the same order as described by the Aufbau principle. f orbital contains 7 boxes that can hold a maximum of 14 electrons.d orbital contains 5 boxes that can hold a maximum of 10 electrons.p orbital contains 3 boxes that can hold a maximum of 6 electrons.s orbital contains 1 box that can hold a maximum of 2 electrons.Let’s see.Įach box will hold a maximum of 2 electrons with opposite spins. These orbitals contain a number of boxes that can hold a number of electrons. There are different types of orbitals – s, p, d, and, f. If you understand the above rules then constructing the orbital diagram or orbital notation for any atom is super easy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
